1Cátedra de Anatomía Comparada, Departamento de Biología, Bioquímica y Farmacia, Universidad Nacional del Sur, B8000CPB, Bahía Blanca, Argentina
2Instituto de Ciencias Biológicas y Biomédicas del Sur (INBIOSUR), Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), B8000ICM, Bahía Blanca, Argentina
3Cátedra de Fisiología Animal, Departamento de Biología, Bioquímica y Farmacia, Universidad Nacional del Sur, B8000CPB, Bahía Blanca, Argentina
For centuries tissue conservation has been sought. Now alternative techniques that minimize the risk of toxicity have emerged. Plastination, developed by Prof. Gunther von Hagens in 1977 is such. The principle of this technique is replacement of the fluid and lipid present in biological tissues by polymers, obtaining odorless, dry, durable and non-toxic specimens. Given these widely recognized benefits, it is an alternative already used by more than 300 scientific-educational institutions around the world. However, the technique requires a laboratory that meets certain structural characteristics, related to biosafety. Construction and start-up involve significant costs. The main objective of this work is to report our experience at Universidad Nacional del Sur (Argentina), where a pre-existing space was modified to transform it into a plastination lab, with substantially lower costs than designing and building from scratch would have required.
anatomical techniques; lab set-up; plastination laboratory; security; soft tissue conservation
Lic. Albertina I. Popp, Departamento de Biología, Bioquímica y Farmacia, Universidad Nacional del Sur, San Juan 670, B80000ICN, Bahía Blanca, Argentina. Tel.: +54 291 4595101, Interno: 2444; E-mail: albertina.popp@uns.edu.ar
![]()



The conservation of soft tissues is a complex task, because it is necessary to use chemical agents that cause some undesirable tissue alterations (loss of color or shrinkage) and are toxic. Among the different substances that can be used as preservative agents, formalin (i.e., aqueous formaldehyde solution) is preferred because it guarantees durability; however, it may cause human health issues (Godish, 1990; Noisel et al., 2007; Whitehead and Savoia, 2008; Dorairajan, 2010; Schwilk et al., 2010; Latorre et al., 2011; Raharyaningsih et al., 2018; Thetkathuek et al., 2020; Adamović et al., 2021). This situation has motivated the search for alternative techniques that help the process of conservation by minimizing the risk of toxicity. Within this framework, the plastination technique was developed (von Hagens, 1979), whose underlying principle is the replacement of organic fluids and tissue fats by harmless polymers (von Hagens et al., 1987). Thereby, through a four-stage procedure (fixation, dehydration, forced vacuum impregnation and curing), odorless, dry, durable, non-toxic materials are obtained, which can therefore be manipulated without the requirement of protective equipment. Because of its widely recognized benefits (Prasad et al., 2015), it represents an advanced solution, currently used by more than 300 scientific-educational institutions in the world (Sora, 2016), and already practised in several South American countries (Bula Calderón, 2012; Muñetón Gómez and Ortiz 2012; Miranda Solis, 2015; Ottone et al., 2015, 2016, 2018; Peralta Pineda, 2017, among others).
Although plastinated specimens are not dangerous, the type of chemicals used during the production process requires that biosecurity measures must be considered. The most critical procedure is dehydration and degreasing of the specimens, since the best solvent for this purpose is acetone, a flammable liquid which gives off a flammable vapor. So, a main concern in designing a plastination lab is to control the risk of fire or explosion. Therefore, to prevent the accumulation of flammable vapors, the space must have an adequate air extraction system, equipped with filters that capture organic vapors to avoid environmental pollution (Beltrán Guerra, 2010; Schill, 2018). During the plastination procedure, acetone must occasionally be handled in open containers; for this reason, and even if the air extraction system works properly, it is necessary to avoid all possible sources of ignition or explosion. This means that electrical circuits (plugs, outlets, switches, and motors) should be explosion-proof, or located outside the laboratory. All these requirements result in high construction costs, and therefore the adaptation of the existing facilities is an alternative that has been implemented elsewhere (Reina de la Torre et al., 2004; Ottone et al., 2014; Zerlotini et al., 2020). The objective of this work is to report our experience at the Universidad Nacional del Sur, starting from the modification of a pre-existing space to transform it into a laboratory suitable for the implementation of the plastination technique with silicones at room temperature.
The Universidad Nacional del Sur (UNS) is located in the central-eastern part of Argentina, at the south of the province of Buenos Aires (38° 44' S; 62° 16' W). The facility, located away from the classroom complex, occupies a total area of ~15 m2, subdivided into two adjoining rooms (Fig. 1A). The largest room was used as a
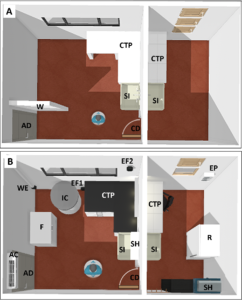
Figure 1. Ceiling view of 3D rendering designs of original space (A) and of the planned modifications (B). AC: air conditioner; AD: access door; CD: communicating door; CTP: countertop; EF1: electric fan at 0.60 m from the floor; EF2: electric fan at 1.80 m from the floor; EP: electrical control panel; F: chest freezer; IC: impregnation chamber; R: refrigerator; SH: shelf; SI: sink; W: wall; WE: air vent with a wind air extractor
conventional laboratory and had an L-shaped tiled countertop with sink. Next to the entrance door, a low dividing wall delimited a corridor, which reduced the useable space and hindered movement. The other room was used for storage, but it also had a countertop and a sink. Both rooms had hot and cold running water and conventional electrical circuits. Considering the biosafety recommendations associated with the technique (Holladay et al., 2001; Schill, 2018, 2019), the largest room was destined for the plastination laboratory and other anatomical techniques, and the smaller used for storage of non-hazardous materials.
Modifications were planned, taking into account the fact that silicones would be used at room temperature. The building changes planned for the plastination room were: removal of the dividing wall, modification of the electrical circuit (rewired, removal of all existing plug in and light keys, and installation of LED lights), installation of air extractors with filtering system, change of the access door for a safety one, and making the countertop sanitary (Fig. 1B). Equipment to be acquired included: chest freezer, refrigerator, impregnation chamber, and furniture (such as shelves, side tables and desk chairs). The construction of an external booth for the motors was designed.
Both the planning and the entire process were supervised by the Occupational Hygiene and Safety Service of our institution (Servicio de Higiene y Seguridad en el Trabajo; Departamento de Biología, Bioquímica y Farmacia; UNS).
Modification of the facilities began in January 2020, with an estimated completion time of eight months. Given the strong and prolonged restrictions due to COVID, the total time was much longer than expected (25 months). Completion was February 2022. The total investment to set up the laboratory was US$7500 (Table 1).
Table 1. Plastination lab modifications and equipment costs
| Modifications and equipment | Cost (US$) |
| Adaptation of the plastination laboratory | 1500 |
| Modification of the electrical circuit | 450 |
| Air extractors | 350 |
| Installation of air extractors | 300 |
| Chest freezer | 500 |
| Technical modifications of freezers | 300 |
| Impregnation chamber | 1300 |
| Furniture | 550 |
| Vacuum pump | 200 |
| Air conditioner | 700 |
| Acetonometers | 50 |
| Generic laboratory equipment | 800 |
| Plastic buckets and other plastic material for laboratory | 500 |
| Total | 7500 |
The modifications resulted in an area of 14.8 m2, between two rooms, one of 9 m2 for the laboratory, and the other 5.8 m2 intended for the storage room along with a refrigerator and necessary furniture (shelves, desk chair and side table) that were installed (Figs. 2 and 3).
All the original electrical circuitry was removed from the entire site and new wiring, explosion-proof LED lights, and touch keys were installed. All the electrical outlets were installed outside the laboratory room. As an extra measure of protection, an electrical control panel with independent thermal keys and circuit breakers (one for each device and circuit), was installed in the storage unit (Fig. 3). In the laboratory room, the dividing wall was removed, and the entrance door was changed for a hermetic, aluminum door, with an anti-panic bar (Fig. 2). A hot and cold air conditioner with temperature control was installed, and the countertop was covered with stainless steel to facilitate cleaning and disinfection. To ensure proper removal of toxic vapors, generated by acetone and other chemical agents, two electric fans (flow: 200 m3/hour, at 1.8 m and 0.6 m above the floor respectively) and an air vent (flow: 100m3/hour, at 0.60 m above the floor) were installed. All air handling units were equipped with an organic vapor filtering system (3M™ Organic Vapor/Acid Gas/Filter).
For the dehydration phase, a 205 L chest freezer, and solvent containers (capacity: 20 liter, D.O.T. carboy) for the acetone, were acquired. A 0.30 m3 cylindrical vacuum chamber (30 cm diameter x 100 cm tall) was built of iron tubing and coated with epoxy paint. A 2 cm-thick toughened glass lid was selected. Both the chest freezer motor and the vacuum pump motor were installed in a special cabinet outside the building, with underground electrical and vacuum line connections.

Figure 3. Storage room. Arrow: electrical control panel containing the thermal keys and circuit breakers
In addition to the described modifications, generic laboratory equipment (instruments, glassware, scales, and various containers) was acquired, as well as supplies for plastination (aquarium pump and two acetonometers).
All the construction and improvements carried out allowed us to start the plastination process with organs from Wistar rats (heart, kidneys, and testicles). For the final disposal of waste products, both liquid (such as acetone, alcohol, and formol) and solid (biological waste), were stored in appropriated containers (biological waste in red biohazard bags, and liquids in waste containers), and collected weekly by the personnel of the Occupational Hygiene and Safety Service of the Universidad Nacional del Sur (https://www.uns.edu.ar/contenidos/64/967#retiro-de-residuos).
The undergraduate and postgraduate courses of the UNS associated with the natural sciences (Biology, Pharmacy, and Biochemistry Medicine) depend on a large number of specimens preserved in formalin. In recent years, efforts have been made to replace this preservative with less harmful ones (isopropyl alcohol), but this has only been possible with the small-sized samples.
Among the existing methodologies to replace or reduce the use of formaldehyde, plastination emerged as a valuable option, since it allows creating quality specimens which are durable and safe, for manipulation by students and teachers (Latorre & López Albors, 2015). However, the construction of a plastination laboratory that meets biosecurity requirements, demanded by the technique, is linked with high costs and a long time for start-up. Our experience and others, demonstrates the possibility to reduce both factors by modifying a pre-existing space (Reina de la Torre et al., 2004; Ottone et al., 2014; Zerlotini et al., 2020). However, since acetone is used in large quantities in the plastination process, it is essential to bear in mind that the process does not only involve a re-adaptation of the space, but certain security requirements must be taken into consideration (Schill, 2018), such as:
The progress we have achieved with the installation of the Plastination Laboratory at the UNS correlates with the need for finding a method of preservation of organic tissues with minimal toxicity, as well as using a room-temperature impregnation technique. The samples generated in this way will be added to our collections to complement the traditional collection of hard tissues (bones, skins, teeth, horns, and shells), which are used both in research and teaching. The availability of these materials will reduce the number of animal cadavers needed for teaching and eliminate human exposure to hazardous substances. The manipulation of these products without the need for gloves is also presented as a promising tool in teaching blind students, who depend on touch as a fundamental sense of information.
Acknowledgments
Fundings: PIP-CONICET 11220200101668CO; PICT-2020-SERIEA-03298, PGI 24/B332, Programa Prombio IF-2016-00614844-APN-SECPU#ME. Additional financial support came from the Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET) through a PhD fellowship to A.I.P.
Adamović D, Cepić Z, Adamović S, Stošić M, Obrovski B, Morača S, Vojinović Miloradov M. 2021: Occupational exposure to formaldehyde and cancer risk assessment in an anatomy laboratory. Int J Environ Res Public Health 18: 11198. https://doi.org/10.3390/ijerph182111198
https://doi.org/10.3390/ijerph182111198
Beltrán Guerra JA. 2010: La plastificación en la Universidad Nacional de Colombia - Primera parte [Plastination at the National University of Colombia - Part I]. Morfolia 2: 3-17.
Bula Calderón AF. 2012: Elaboración de material docente mediante la utilización de la técnica de plastinación en modelos formolizados como método ideal de enseñanza para los Estudiantes de Medicina de la Universidad Nacional de Colombia [Preparation of teaching material through the use of the plastination technique in formalized models as an ideal teaching method for Medical Students of the National University of Colombia]. MSc Thesis. Universidad Nacional de Colombia, Bogotá, 129 pp.
Dorairajan G. 2010: Formalin: Nephrotoxic teratogen? J Obstet Gynaecol Res 36: 1256-1260.
https://doi.org/10.1111/j.1447-0756.2010.01309.x
Godish T. 1990: Residential formaldehyde: Increased exposure levels aggravate adverse health effects. J Environ Health 53: 34-37.
Holladay SD, Blaylock BL, Smith BJ. 2001: Risk factors associated with plastination: I. Chemical toxicity considerations. J Int Soc Plast 16: 9-13.
https://doi.org/10.56507/CWZW6925
Latorre R, López Albors O. 2015: Plastination. Eur J Anat 19: 39-45.
Latorre N, Silvestre J, Monteagudo A. 2011: Allergic contact dermatitis caused by formaldehyde and formaldehyde releasers. Actas Dermosifiliogr 102: 86-97.
https://doi.org/10.1016/S1578-2190(11)70765-X
Miranda Solis F. 2015: La plastinación como método de conservación de tejidos biológicos para docencia e investigación en la anatomía humana [Plastination as a method of preservation of biological tissue for teaching and research in human anatomy]. Rev Peru Med Exp Salud Pública 32: 819-820.
https://doi.org/10.17843/rpmesp.2015.324.1780
Muñetón Gómez CA, Ortiz JA. 2012: Plastinación: un instrumento complementario para el desarrollo del proceso enseñanza-aprendizaje de la anatomía [Plastination: A complementary tool for the development of the teaching-learning process of anatomy]. Rev Med Vet 23: 111-117.
https://doi.org/10.19052/mv.79
Noisel N, Bouchard M, Carrier G. 2007: Evaluation of the health impact of lowering the formaldehyde occupational exposure limit for Quebec workers. Regul Toxicol Pharmacol 48: 118-127.
https://doi.org/10.1016/j.yrtph.2007.02.001
Ottone NE, Bianchi HF, Fuentes Fernandez R, Aja Guardiola S, Cirigliano V, Oloriz L, Borges Brum G, Blasi E, Algieri RD, Bertone VH. 2014: Development and installation of a plastination laboratory using low-cost equipment and materials. Abstracts 17th International Conference on Plastination, Saint Petersburg, Russia, 14-18/07/2014. https://journal.plastination.org/my_abstracts/abstracts-from-the-17th-international-conference- on-plastination-saint-petersburg-russia-july-14-18-2014
Ottone NE, del Sol M, Fuentes R. 2016: Report on a sheet plastination technique using commercial epoxy resin. Int J Morphol 34: 1039-1043.
https://doi.org/10.4067/S0717-95022016000300036
Ottone NE, Baptista C, Latorre R, Bianchi HF, Del Sol M, Fuentes R. 2018: E12 sheet plastination: Techniques and applications. Clin Anat 31: 742-756.
https://doi.org/10.1002/ca.23008
Ottone NE, Cirigliano V, Bianchi HF, Medan CD, Algieri RD, Borges Brum G, Fuentes R. 2015: New contributions to the development of a plastination technique at room temperature with silicone. Anat Sci Int 90: 126-135.
https://doi.org/10.1007/s12565-014-0258-6
Peralta Pineda E. 2017: Aplicación de la técnica de plastinación en órganos humanos utilizando látex en la generación de modelos anatómicos para la enseñanza de la morfología humana [Application of the plastination technique in human organs using latex in the generation of anatomical models for the teaching of human morphology]. MSc Thesis. Universidad Nacional de Colombia, Bogotá, 95 pp.
Prasad G, Karkera B, Pandit S, Desai D, Tonse RG. 2015: Preservation of tissue by plastination: A review. Int J Adv Health Sci 1: 27-31.
Raharyaningsih MA, Azizah R. 2018: Air formaldehyde levels and eye irritation in workers at production area of wood adhesive factory in Surabaya. J Kesehat Lingk 9: 191-199.
https://doi.org/10.20473/jkl.v9i2.2017.191-199
Reina-de La Torre F, Rodríguez-Baeza A, Doménech-Mateu JM. 2004: Setting up a plastination laboratory at the Faculty of Medicine of the Autonomous University of Barcelona. Eur J Anat 8: 1-6.
Schill VK. 2018: General issues of safety in plastination. J Plast 30: 27-36.
https://doi.org/10.56507/NJCY9228
Schill VK. 2019: Work safety in plastination. Anat Histol Embryol 48: 584-590.
https://doi.org/10.1111/ahe.12473
Schwilk E, Zhang L, Smith MT, Smith AH, Steinmaus C. 2010: Formaldehyde and leukemia: An updated meta-analysis and evaluation of bias. J Occup Environ Med 52: 878-886.
https://doi.org/10.1097/JOM.0b013e3181ef7e31
Sora MC. 2016: The general protocol for the S10 technique. Res Clin Med 1: 14-18.
Thetkathuek A, Yingratanasuk T, Ekburanawat W, Jaidee W, Sa-Ngiamsak T. 2020: The risk factors for occupational contact dermatitis among workers in a medium density fiberboard furniture factory in Eastern Thailand. Arch Environ Occup Health 76: 255-265.
https://doi.org/10.1080/19338244.2020.1819185
von Hagens G. 1979: Impregnation of soft biological specimens with thermosetting resins and elastomers. Anat Rec 194: 247-255.
https://doi.org/10.1002/ar.1091940206
von Hagens G, Tiedemann K, Kriz W. 1987: The current potential of plastination. Anat Embryol 175: 411-421.
https://doi.org/10.1007/BF00309677
Whitehead MC, Savoia MC. 2008: Evaluation of methods to reduce formaldehyde levels of cadavers in the dissection laboratory. Clin Anat 21: 75-81.
https://doi.org/10.1002/ca.20567
Zerlotini MF, Paula TAR, Ramos ML, Silva FFR, Santana ML, Figueira MP, Silva LC, Silva VHD, Bustamante LRC. 2020: Establishment and operationalization of a low-budget plastination laboratory in the Veterinary Morphology Section of the Federal University of Viçosa, Minas Gerais - Brazil. J Plast. 32: 8-17.
https://doi.org/10.56507/RKZU4777