1 National Museum of Underwater Archaeology, Cartagena, Spain
2 Department of Anatomy and Comparative Pathological Anatomy; Regional Campus of International Excellence “Campus Mare Nostrum”, University of Murcia, Spain
The archaeological excavation of a Phoenician shipwreck at Bajo de la Campana, San Javier, Murcia, Spain, was carried out from 2007 to 2011 under a collaboration agreement between the Institute of Nautical Archaeology of Texas A & M University, and the Ministry of Culture of Spain1, through the National Museum of Underwater Archaeology of Cartagena.
Throughout successive campaigns of systematic excavation, the archaeologists documented and raised an extraordinary cargo of a Phoenician wreck dated between 7th – 6th centuries BC. Among the raw materials it carried there was a magnificent set of 53 elephant tusks, and fragments of elephant tusks, some of them with inscriptions.
The uniqueness of this archaeological find is due to it being one of the few known examples of Phoenician navigation in the Mediterranean Sea, and the first time a shipment carrying ivory as raw material has been documented in Spain.
With this work, we present the results of the plastination process carried out on waterlogged archaeological remains, as an alternative to traditional preservation procedures on items of cultural heritage, which have proved to be ineffective due to the extreme density of ivory.
We tested the Biodur® S15 plastination technique at room temperature in successive phases, each step depending on the results obtained in the previous steps. Elephant tusk fragments were dehydrated with acetone, and subsequently impregnated with a mixture of polymer and catalyst, Biodur® S15 plus 1% S3. After impregnation, they were exposed to Biodur® S6 crosslinker, for the curing phase. The time taken for each step varied proportionally, according to the size of the ivory fragments.
The results obtained were satisfactory, both in dimensional stability and visual aspect, essential for the study and exhibition of these pieces that are part of our cultural heritage. It should be noted that these optimum results have been maintained over time, two years since their processing, which validates this technique of plastination as a procedure for preserving waterlogged archaeological materials.
1 Ministry of Education, Culture and Sport, Secretary of State for Culture since December 2011
archaeology; conservation; ivory; plastination; Biodur©S15; waterlogged
M. BUENDÍA1, 30202 Cartagena, Murcia, Spain. Tel.: Office: +34 968 98 15 62; Email: milagros.buendia@mecd.es
![]()



The conservation of artifacts of our cultural heritage is based on a sound knowledge of the material’s alteration factors, and the deterioration processes suffered during the burial period. In this case, it was necessary to carry out an initial characterization of archaeological waterlogged ivory, and compare it with samples of non-degraded ivory 2. This enabled us to understand the structural components and diagenetic processes due to the marine environment.
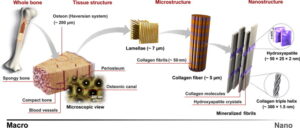
Figure 1: Hierarchical structure of the structural components of bone and ivory (Sadat-Shojai et al., 2013, p.7592, Figure 1)
Ivory is composed of a combination of organic and inorganic components, calcium hydroxyapatite and collagen type I, which are intimately related in a hierarchical way, from nano- to macro-scale, that gives ivory its extraordinary physico-mechanical properties (Fig. 1).
The main diagenetic processes that the ivory had undergone during its burial period were partial dissolution of its structural components (mainly the organic fraction), and incorporation of exogenous elements that had replaced ions present in the hydroxyapatite structure, increasing the degree of crystallinity. In the case of iron, these had precipitated forming inclusions of pyrites that occupied fissures and interstices in
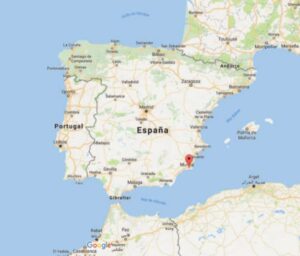
Figure 2: Location of Bajo de la Campana site, opposite the Mar Menor, Murcia (Image taken from Google Earth and modified)
the apatite matrix (Doménech-Carbó et al., 2016). As a result, we had elephant tusks that were highly degraded, and in which water had become part of the structure of the ivory, with a clear supporting function, so that its loss would cause internal stresses that would cause the structure of the ivory to collapse irreversibly. We carried out a study of waterlogged ivory degradation, and the damage suffered, on an archaeological ivory section from a Phoenician shipwreck (7th-6th centuries BC) at Bajo de la Campana3 (Figs. 2, 3), subjected to natural drying, without any conservation treatment. The section experienced a total weight loss of almost 44%. The most important changes occurred on the cross section, however, a few smaller changes were also observed in tangential and longitudinal sections, due to the anisotropy of the ivory (Fig. 4 i-iii).
As part of Spain’s cultural heritage, our priority was to preserve these items, in order to
be able to access the historical and scientific information they hold. For this reason, it was necessary to apply a conservation procedure that allowed us to eliminate water, as well as guaranteeing dimensional stability of the artifacts.
The conservation of underwater cultural heritage begins with the implementation of a planned program of actions, from the initial planning of the archaeological intervention, taking in to consideration human resources, materials, and infrastructures, both during the work at the site, and throughout the raising work, especially with the most fragile materials; and to then take them to the museum’s conservation laboratory, where the gradual transition from the marine environment to dry conditions will take place.
All artifacts that come to the conservation laboratory are individually inventoried and documented. In this way we record the initial conservation state and all necessary subsequent conservation treatments.
| Figures 4i – 4III: Sequence of damage observed during the drying phase of the archaeological elephant tusk section SJBC_11_2471_3 (MNAS photographic archive) | ||
The conservation work began by removing soluble salts, by successive fresh water and demineralized water baths, to avoid further recrystallization and irreversible damage. The tusks were then prepared for drying, however, in the case of waterlogged materials, the degradation they would suffer made natural drying impossible. It was necessary to apply a conservation process that removes water, reinforces damaged structure, and guarantees dimensional stability of the artifact. The process must be compatible with the ivory and its exogenous components, (either from the marine environment, or neo-formed), that cannot be eliminated.
The Biodur® S10 and Biodur® S15 plastination procedures (von Hagens, 1988), originally developed and patented by Dr. Gunther von Hagens in the late 1970s for the preservation of organic specimens for anatomy teaching, provides characteristics that make it suitable for our purpose, such as chemical stability of its components, and good diffusion (Whalley, 1988; Wade and Lyons, 1995, 1999).
Ian Godfrey from The Western Australian Museum carried out plastination with Biodur® S10 on fragments of ivory from a Dutch merchant ship, the Vergulde Draeck (1656) obtaining optimal results at room temperature (Godfrey et al., 2012).
Other companies such as Silicone Inc., Corcoran Chemicals® or Dow Corning® have formulated very fluid silicone polymers that can be used at room temperature, with slightly modified procedures: the North Carolina room-temperature plastination technique, and the Dow®/Corcoran technique (Raoof et al., 2007). The Archaeological Preservation Research Laboratory at Texas A&M University has worked with highly fluid polymers4 that can be applied at room temperature with almost no vacuum (silicone oil preservation) (Smith, 2003), which are more suitable conditions for conservation of archaeological artifacts. Wayne Smith has reported excellent results obtained with organic and inorganic materials from the wreck of La Belle (17th century), waterlogged ivory from the Tantura B shipwreck, (9th century), and from the wreck of the merchant ship the Vergulde Draeck (17th century).
We applied the Biodur® S15 plastination procedure at room temperature because we needed to dry and reinforce damaged archaeological ivory from Bajo de la Campana, and we lacked a detailed, easily reproducible plastination protocol. The most common protocol used in plastination laboratories is Biodur® S10 and Biodur® S15, although a slight modification of the latter technique was required for waterlogged archaeological ivory.
We proposed as the aims of this study, the development of a specific protocol for the Biodur® S15 plastination technique at room temperature, which would allow a better diffusion in an extremely dense material such as ivory, and enable not only the assessment its effectiveness on this type of material, but also the long-term stability of the treatment, through validation over time.
|
|
2 Artifact with Community Certificate number ESAA0000813C comes from a forfeiture for breach of the Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES) and the Implementing Regulations in the European Union.
3Tusk fragment section, nº inv. SJBC_11_2471_3, dimensions and initial weight: Cross section: 5cm x 4 cm, thickness: 4.7 cm, waterlogged weight: 80.28 g.
4Dow Corning®: PR-10 hydroxyl-ended, functional polymer, CR-20 crosslinker (methyltrimethosysilane/siloxaneQ1-3563), CT-32 catalyst (dibutyltin diacetate)
Application of the plastination procedure, Biodur® S15 method, at room temperature
Experiments / trials
The application of the Biodur® S15 plastination procedure was carried out in progressive steps, each depending on the results obtained in the previous steps.
Stage 1: To define a specific plastination protocol for an ivory specimen. It was performed on an elephant tusk section, the first time the procedure was applied to an archaeological artifact, specimen SJBC_11_2471_5a (Fig. 5). The specimen’s dimensions and weight before
Stage 2: Validate the protocol used in Stage 1 with an elephant tusk fragment, to verify the effectiveness of the procedure, and to establish if the shape and volume of the fragment will influence the behavior of the plastination process. The stability achieved in Stage 1, with the room temperature Biodur® S15 procedure, led us to investigate if equally good results could be achieved with a tusk fragment, that had not been sectioned. For this trial, we selected a tusk fragment with a preserved distal end, the proximal part having been mostly lost, specimen SJBC_10_2980 (Fig. 6). The specimen’s dimensions and weight before treatment were: straight line length between proximal and distal ends 22 cm, outer curvature 25 cm, maximum diameter 4.6 cm, waterlogged weight 373.61 g.
Stage 3: The validated protocol was applied to a complete elephant tusk (SJBC_10_1926), for confirmation of the previous encouraging results, and the long-term study of the effectiveness of the treatment. The good results achieved on tusk section SJBC_11_2471_5a and on tusk fragment SJBC_11_2980, encouraged us to apply the S15 plastination procedure to a complete elephant tusk, SJBC_10_1926 (Fig. 7). The tusk dimensions were: straight line length between proximal and distal ends 72 cm, outer curvature 100 cm, maximum diameter 8.1 cm, waterlogged weight 4.850 kg.
The phases of the Biodur® S15 procedure at room temperature were, as in the standard protocol, dehydration, impregnation, and curing. Specimens were dehydrated in successive acetone baths, giving final acetone concentrations from 98.5% to 100%. Dehydration was carried out in a refrigerated chamber, at +5 ° C, to avoid contraction and tension forces.
Impregnation was carried out at room temperature (18-20° C) in a vacuum chamber, with the impregnation mixture of silicone polymer Biodur® S15 mixed with the chain-extender S3 (1%). The impregnation chamber pressure during impregnation was gradually decreased from 760 mm Hg to 3 mm Hg. Continuous bubbling was observed, as a consequence of acetone exit, and the progressive incorporation of polymer into the specimens. Impregnation was considered finished when no further bubbles appeared on the surface of the silicone bath. The vacuum was released, and the tusks remained in the impregnation bath overnight. The specimens were then taken out of the bath, and excess polymer was removed with cellulose tissue paper. Subsequently, the specimens were placed in a curing chamber where cross-linker (Biodur® S6) was vaporized to saturate the interior and cause curing to occur.
The chemicals used in this procedure were acetone, Biodur® S15 polymer, Biodur® S3 catalyst, and Biodur® S6 curing agent (von Hagens et al., 1988; Henry et al., 1997; Latorre et al., 2001; Smodlaka et al., 2005; Ottone et al., 1988).
All the archaeological ivory sections, tusk fragments and complete elephant tusks were documented before and after plastination, photographed, measured, and weighed, to record any changes that occurred during processing.
Table 1 shows the time taken for the three plastination stages for each specimen. Table 2 shows the weight of the specimens, from the initial to the final weight, and percentage weight loss.
Specimen 1: section nº 5a from tusk fragment SJBC_11_2471_5a (Figs. 8-11).
The total plastination time for this specimen was 9 days (Table 1). After dehydration with acetone, an incomplete crack was observed.
| Inventory number | DEHYDRATION Acetone5 °C |
IMPREGNATION S15+S1
Room temp. |
CURING S6Room temp. |
| SJBC_11_2471_5a (piece 1) | 3 days | 3 days | 3 days |
| SJBC_11_2980 (piece 2) | 27 days | 14 days | 12 days |
| SJBC_10_1926 (piece 3) | 27 days | 30 days | 12 days |
The total plastination time for this specimen was 9 days (Table 1). After dehydration with acetone, an incomplete crack was observed. During impregnation,

Figure 12. Tusk section SJBC_11_2471_5a cured (Laboratory of Plastination, Faculty of Veterinary Medicine, University of Murcia)
no incidents were detected, and the crack did not increase. When the tusk section was exposed to cross-linker, it began to change color until the drying was complete. The final appearance was very similar to dry ivory; no glossy deposits of polymer were seen on the surface or interstices (Fig. 12).
After curing, the weight of the tusk section was monitored, and it was found to decrease from 46.72 g to 44.01 g at three months. The fissure remained stable, it did not progress, and no new cracks appeared. The difference between initial and final weight was 5.55 g, 11.32% of its initial weight (Table 2).
| Inventory number |
Waterlogged initial weight (grams) |
Final weight (grams) |
Final weight (grams) |
Final weight (grams) |
Weight lost
|
| SJBC_11_2471_5a (Piece 1) | 49.01 | 46.72 | 44.01 | 43.46 | 11.33 |
| SJBC_11_2980 (Piece 2) | 373.61 | 370.70 | 365.75 | 365.96 | 2.05 |
| SJBC_10_1926 (Piece 3) | 4850.0 | 4445.0 | 4445.0 | 4443.28 | 8.61 |
Table 2. Weight of ivory pieces before and after plastination, and percentage weight loss
The complete process for this specimen took 53 days (Table 1). During dehydration, no fissure, crack, or detachment was observed, and the tusk fragment remained stable. After impregnation, the excess polymer was not removed; on the contrary, more impregnation mixture (S15 + S3 1%) was applied to the surface, to determine if leaving this thick layer on the specimen was more effective in preventing possible loss or detachment of cementum or dentin. It also allowed us to investigate how to remove excess surface polymer during curing, and if, in doing this, any loss of material occurred (Fig. 13).
The final weight of the plastinated tusk fragment was 365.96 g, weight loss 2.05% (Table 2).
Removal of the curing polymer from the surface of tusk fragment SJBC_11_2980 was carried out using mechanical procedures. We used a scalpel, by scraping the superficial layer of silicone, and, subsequently, rubbing the residual polymer with a swab (Fig. 14). It was even possible to remove this layer by using a swab alone, by scraping and rubbing with the swab, because the cotton adhered to the silicone and removed it perfectly. It took a little more time but was very controllable and effective. The original surface of the tusk fragment was preserved, so that the original rough appearance can be seen. No detachment of the tusk surface occurred, and the fragment remained stable (Fig. 15).
Specimen 3: Complete elephant tusk SJBC_10_1926 (Figs. 16-19)
The complete plastination procedure took 69 days. During dehydration and impregnation (Figs. 16-18) no fissures, cracks or detachment appeared; the tusk remained stable. After impregnation, the excess polymer was removed from the surface with cellulose tissue paper. This caused local detachments of cementum. A small fragment of the proximal end that had a micro-crack prior
to treatment also became detached (Fig. 19). It appeared that leaving a thick layer of excess polymer protected the tusk surface. The surface-cured polymer was cleaned by mechanical means. By rubbing with a swab, the polymer was removed smoothly (Fig. 19). No new cracks have been detected, nor has there been any progression in existing defects. The final weight was 44432.80 g; weight loss was 8.61% (Table 2).
Waterlogged elephant tusks recovered and documented from archaeological sites around the world are very scarce, which is why little is known about their conservation.
Our objective was conservation of waterlogged elephant tusks from the Phoenician Bajo de la Campana shipwreck, and, in doing so, to enhance our knowledge of the plastination procedure and its application to conservation of cultural heritage. We have, for the first time, documented the Biodur®S15 plastination technique on waterlogged archaeological ivory, and how to remove the excess cured polymer from its surface. Our results provide an easy-to-reproduce protocol for using the Biodur® S15 plastination technique at room temperature.
In previous conservation work on waterlogged archaeological ivory, satisfactory results were obtained with Biodur®S10 and the silicone oil /siloxane method, respectively (Godfrey et al 2012; Smith 2003). Smith used a variation in the standard method, which was impregnation with a mixture of polymer and cross-linker with light vacuum, and curing with catalyst.
Godfrey used Biodur® S10 and siloxane Q1-3563 containing MTMS (9%) (Godfrey et al., 2012), while Smith used SFD-1 (Dow Corning® PR-10) silicone oil containing 5% methyltrimethoxysilane (Dow Corning® CR-20, crosslinker) and dibutylin diacetate (Dow Corning® CT-32, catalyst) on waterlogged archaeological ivory from the Vergulde Draeck shipwreck (17th C) (Smith, 2003). In both cases, the steps used were dehydration with alcohol or acetone, impregnation at room temperature, and curing; this is the basis of all room-temperature protocols, in order to avoid the silicone becoming viscous too quickly from the effects of the catalyst or chain elongator (Ludwikowski et al., 2001; Raoof, 2001).
In our opinion, the critical point of this plastination procedure was to be able to preserve the integrity of the specimen, particularly the most fragile fragments on surface, avoiding any becoming detached. The fissures and detachment that we have documented occurred because we worked with highly degraded material, however, they were extremely useful to improve the procedure. The results obtained in our study show that the Biodur® S15 plastination technique at room temperature, was successfully applied to the Bajo de la Campana ivory.
Dehydration was the stage where we observed some changes, a small crack appeared in specimen 1 (Fig. 8) which seemed to be related to the size of tusk section and the length of time spent in this phase. In addition to this, water plays an important role as a cohesive element in organic and mixed waterlogged archaeological materials.
Other authors who have worked with waterlogged ivory, and other archaeological materials such as waterlogged wood or leather, (De La Cruz Baltazar, 1996; De La Cruz Baltazar et al., 1999; Bouzas et al., 2008), have not reported any limitations during this phase.
The impregnation phase was carried out without problems. The mixture of silicone polymer and catalyst was perfectly incorporated. Bubbling indicated the evaporation of acetone until it ceased; the larger the piece, the longer this phase takes. The end of the process was reached at pressures between 5 mm-2 mm Hg; this did not cause any damage to the archaeological ivory.
An important disadvantage of silicone oil preservation is the partial loss, by evaporation, of cross-linker at pressures below 10 mm Hg during the impregnation phase. This means that there is no control over the percentage of cross-linker remaining in the impregnation mixture.
Tests performed on Specimen 2 showed that it was necessary to keep a layer of silicone on the surface as a stabilizing element for the more fragile lamellae. Subsequently, as we have explained above, it is then necessary to remove part of the protective layer of silicone mechanically, in order to achieve a natural final appearance.
The successful control of both weight loss and shrinkage of the Bajo de la Campana’s ivory pieces shows that a significant dimensional stability can be achieved, a crucial requirement for underwater archaeological heritage conservation. On the one hand, the polymer becomes a supporting element of the damaged structure, replacing the structural components lost or transformed during the immersion period; and, on the other hand, it stabilizes even the most unstable incorporated and neo-formed compounds, such as pyrite (Doménech et al., 2016). The maximum weight loss obtained with the Biodur® S15 plastination technique at room temperature was 11%, however, previous assays in our lab with the classical protocol of monitored desiccation showed a weight loss greater than 40%.
Godfrey et al., (2012) analyzed plastinated ivory fragments ten years after treatment, and reported no apparent changes. They emphasized the high degree of penetration, homogeneous diffusion, and structural reinforcement of ivory specimens with diverse initial states of conservation, both slightly degraded and very degraded. However, these authors do not comment on long-term weight loss.
Our results, two years after treatment, validate Biodur® S15 plastination at room temperature as a conservation method, accomplishing an optimal final appearance. The plastinated ivory has a natural color, and the texture, (whether the excess polymer is removed with cellulose paper after impregnation, or mechanically removed after curing), is practically same as the original.
This plastination procedure achieves the main objectives of water removal and dimensional stability. Plastination is also reversible, a preferred, but not essential, quality in conservation of cultural heritage. It is important to have the possibility of reversing the procedure, in case it causes problems in the future. The process of de-plastination of plastinated ivory could be an area for future study, since there are currently no references to this in the literature (Walker et al., 1988, De La Cruz Baltazar et al., 1999).
In conclusion, plastination can be an effective conservation procedure for waterlogged organic and mixed materials, although we will continue to monitor its long-term outcomes, and continue our research into the conservation of waterlogged ivory.
Bouzas A, de Castro JM, Zambrano LC. 2008: Tratamientos de la madera arqueológica saturada de humedad por el método denominado plastinación. PH, Bol Inst Andal Patrim Hist 65:108-119.
https://doi.org/10.33349/2008.65.2498
De La Cruz Baltazar V, Lyons W, Murray A, Hanlan, J. 1999: Plastination as a consolidation technique for archaeological bone, wet leather and waterlogged wood. 6th Interim Conf Plast, Rochester, NY, USA, 1999. Abstract in J Int Soc Plastination 14:35.
De La Cruz Baltazar, V. 1996: Plastination as a Consolidation Technique for Archaeological Bone, Waterlogged Leather and Waterlogged Wood. Master of Art Conservation Thesis, Queen's University, Kingston, Ontario, Canada, 177 pp.
Doménech-Carbó MT, Buendia Ortuño M, Pasíes Oviedo T, Osete Cortina L. 2016: Analytical study of waterlogged ivory from Bajo de la Campana site (Murcia, Spain). Microchem J 126:381-405.
https://doi.org/10.1016/j.microc.2015.12.022
Godfrey I, Kasi K, Lussier S, Smith, CW. 2012: Conservation of waterlogged elephant tusks. In: Straetkvern K, Williams E, editors. Proceedings of the 11th ICOM Working Group on Wet Organic Archaeological Materials Conference, Greenville 2010. International Council of Museums, p 633-646.
Latorre R, Vázquez JM, Gil F, Ramírez G, López-Albors O, Orenes M, Martínez-Gomariz F, Arenciba A. 2001: Teaching Anatomy of the Distal Equine Thoracic Limb with Plastinated Slices. J Int Soc Plast 16:23-30.
https://doi.org/10.56507/ACRF7155
Ludwikowski B, Oesch Hayward I, Brenner E, Fritsch H: The development of the external urethral sphincter in humans. BJU Int 87 (6): 565-568, 2001.
https://doi.org/10.1046/j.1464-410X.2001.00086.x
Ottone NE, Cirigliano V, Bianchi HF, Medan CD, Algieri RD, Borges Brum G, Fuentes R. 1988: New contributions to the development of a plastination technique at room temperature with silicone. Anat Sci Int 90 (2): 126-135.
https://doi.org/10.1007/s12565-014-0258-6
Raoof A, Henry RW, Reed RB. 2007: Silicone Plastination of biological tissue: room-temperature technique Dow®/Corcoran technique and products, J Int Soc Plast 22:21-25.
https://doi.org/10.56507/AWAC9285
Raoof A. 2001: Using a Room-Temperature Plastination Technique in Assessing Prenatal Changes in the Human Spinal Cord. J Int Soc Plastination 16: 5-8.
https://doi.org/10.56507/UIHR4575
Smith CW. 2003: Archaeological Conservation Using Polymers: Practical Applications for Organic Artefact Stabilization. College Station: Texas A&M University Press.
Smodlaka H, Latorre R, Reed RB., Gil F, Ramírez G, Váquez-Autón JM, López-Albors O, Ayala MD, Orenes M, Cuellar R, Henry RW. 2005: Surface Detail Comparison of Specimens Impregnated using Six Current Plastination Regimens. J Int Soc Plast 20:20-30.
https://doi.org/10.56507/FIHG2408
Von Hagens, G. 1988: Curing of brain specimens in the S-15 technique. 4th Int Conf Plast, Macon, GA, USA.
Wade R, Lyons W. 1995: The restoration of anatomical and archaeological specimens using S-10 plastination method: With special reference to preserving the good heart of a good priest. II World Congress on Mummy Studies, Cartagena, Columbia.
Wade R, Lyons W. 1999: The restoration of anatomical and archaeological specimens using the S10 plastination method: with special reference to preserving the good heart of a good priest. 6th Interim Conf Plast, Rochester, NY, USA, 1999. Abstract in J Int Soc Plast 14 (2): 34.
Walker AN, Jackson RL, Powell S. 1988: Technical communication: routine microscopy of deplastinated tissue. J Int Soc Plast 2 (1): 40-42.
https://doi.org/10.56507/ODUA6186
Whalley A. 1988: Practical applications of plastination in archeology. 4th Int Conf Plast, Macon, GA, USA.